Quadriceps Strength Symmetry and Patient-reported Function Early after ACL Reconstruction

Introduction
Quadriceps weakness is a predictor of worse long-term knee function and strength recovery can vary from months to years after ACL reconstruction (ACLR). Up to now, little is known about quadriceps strength and self-reported function within the first weeks after ACLR. The purpose of this study was to examine quadriceps strength symmetry and knee-related function pre-operatively and at six, 12, and 24 weeks post-ACLR.
Methods
This study recruited individuals with acute primary unilateral ACL ruptures scheduled for ACLR from a larger RCT. ACL rupture was confirmed by a positive Lachman, anterior drawer or pivot shift test, and magnetic resonance imaging. The individuals had to participate in cutting, pivoting, jumping, and lateral movement sports for at least 50 hours per year prior to ACL injury to be eligible candidates.
ACLR was performed with a hamstring autograft and rehabilitation was standardized using ACLR criterion-based rehabilitation guidelines based on the Multicenter Orthopedic Outcomes Network (MOON) recommendations.
Quadriceps strength was measured through isometric and isokinetic dynamometry (pre-ACLR and at 12 and 24 weeks). At 6 weeks isometric strength with the knee fixed in 90° of flexion was used to measure quadriceps strength and to protect the ACL graft from excessive strain. Peak torque was normalized to body weight and used to calculate the quadriceps index (QI). Knee-related function was measured through the Knee injury and Osteoarthritis and Outcome Score (KOOS) and International Knee Documentation Committee Subjective Knee Evaluation Form (IKDC) questionnaires. On both questionnaires, higher scores (0-100) represent higher functional status.
Results
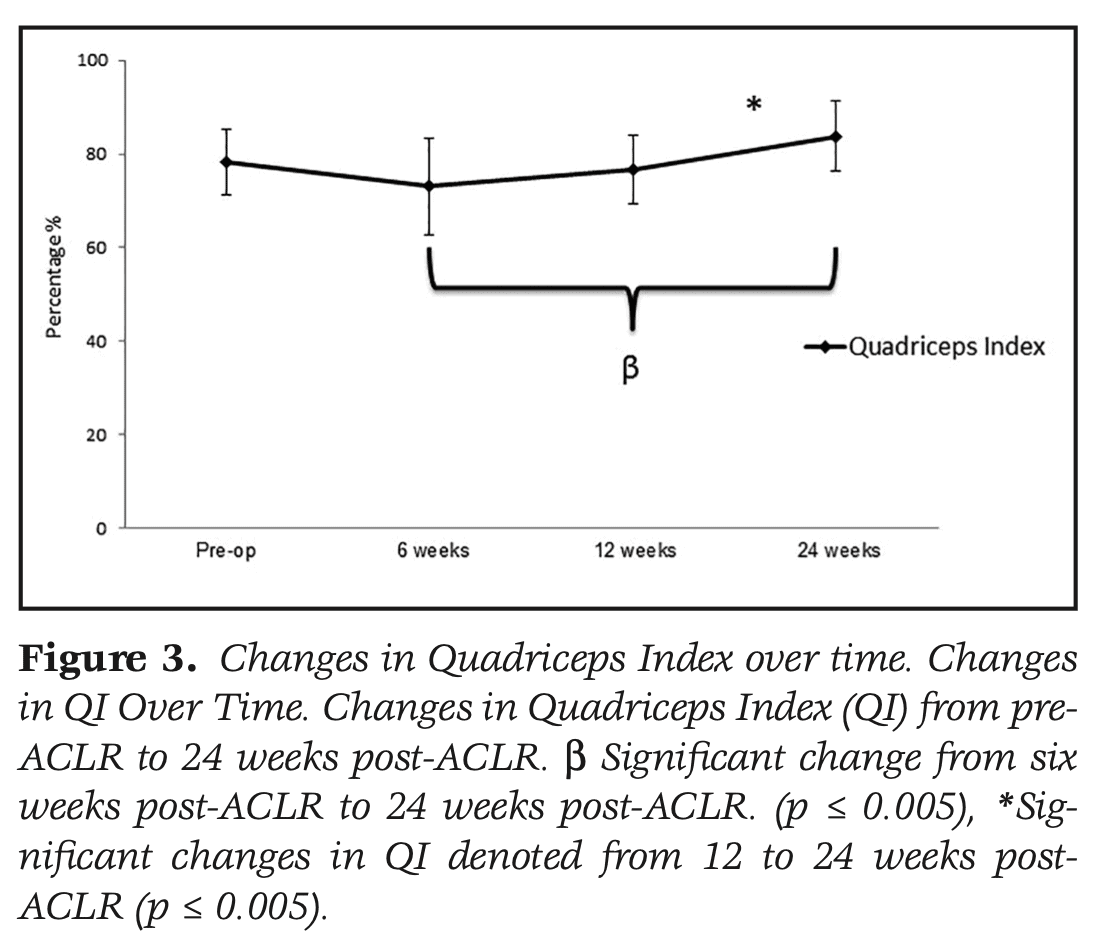
Quadriceps peak torque improved significantly over the course of the study. Significant improvements were seen between 12 weeks and 24 weeks. The QI also improved significantly over time. Between 12 and 24 weeks post ACLR, the QI improved significantly from 74% to 84%.
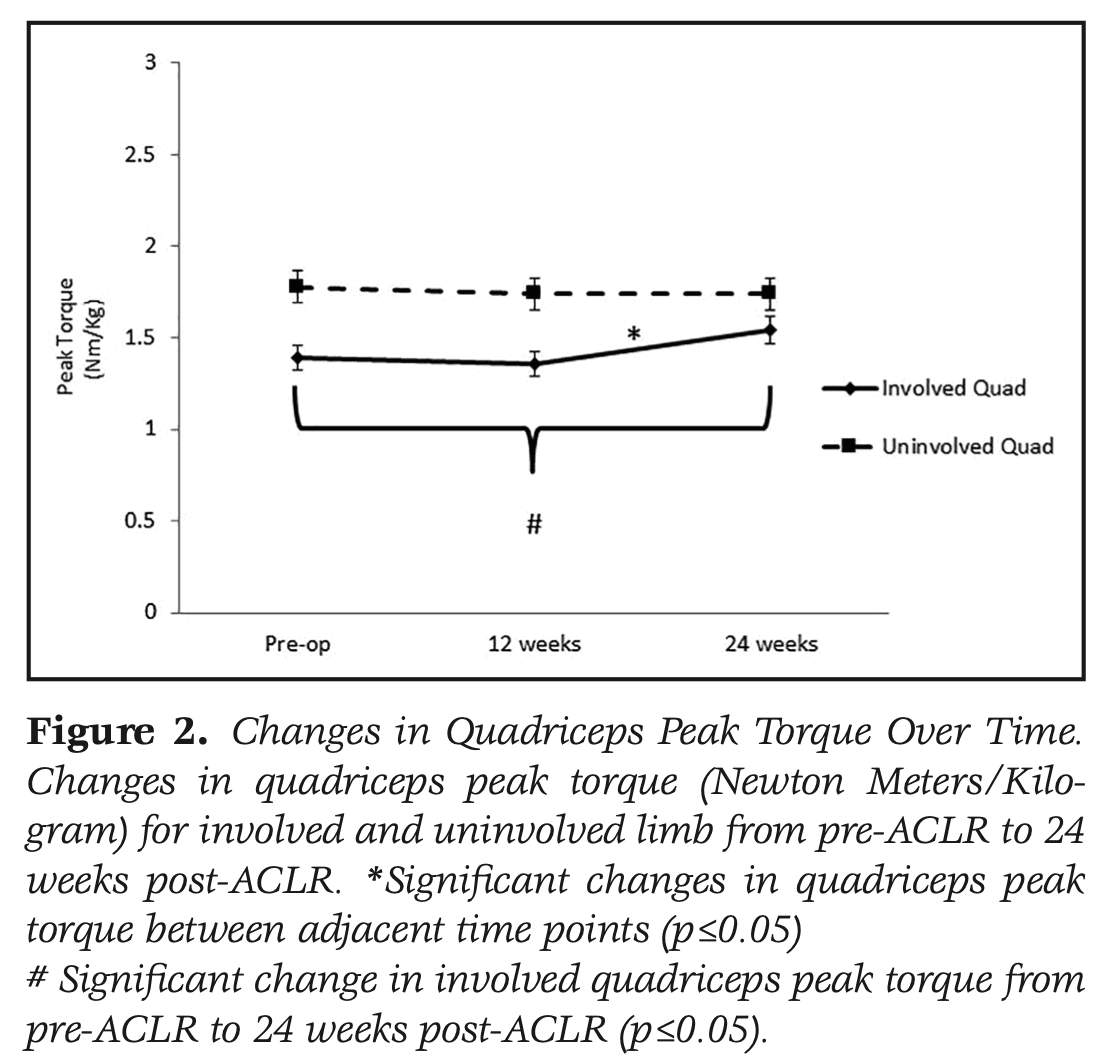
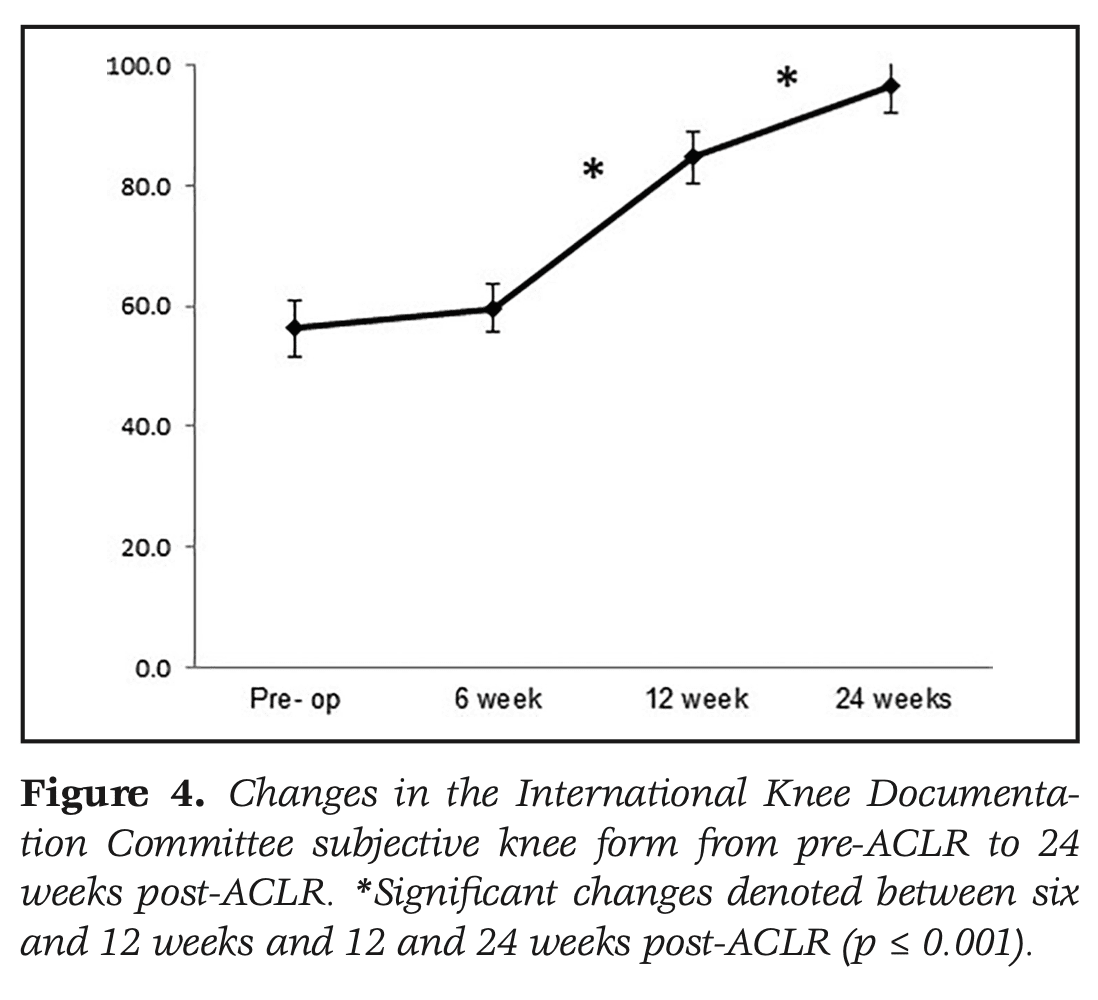
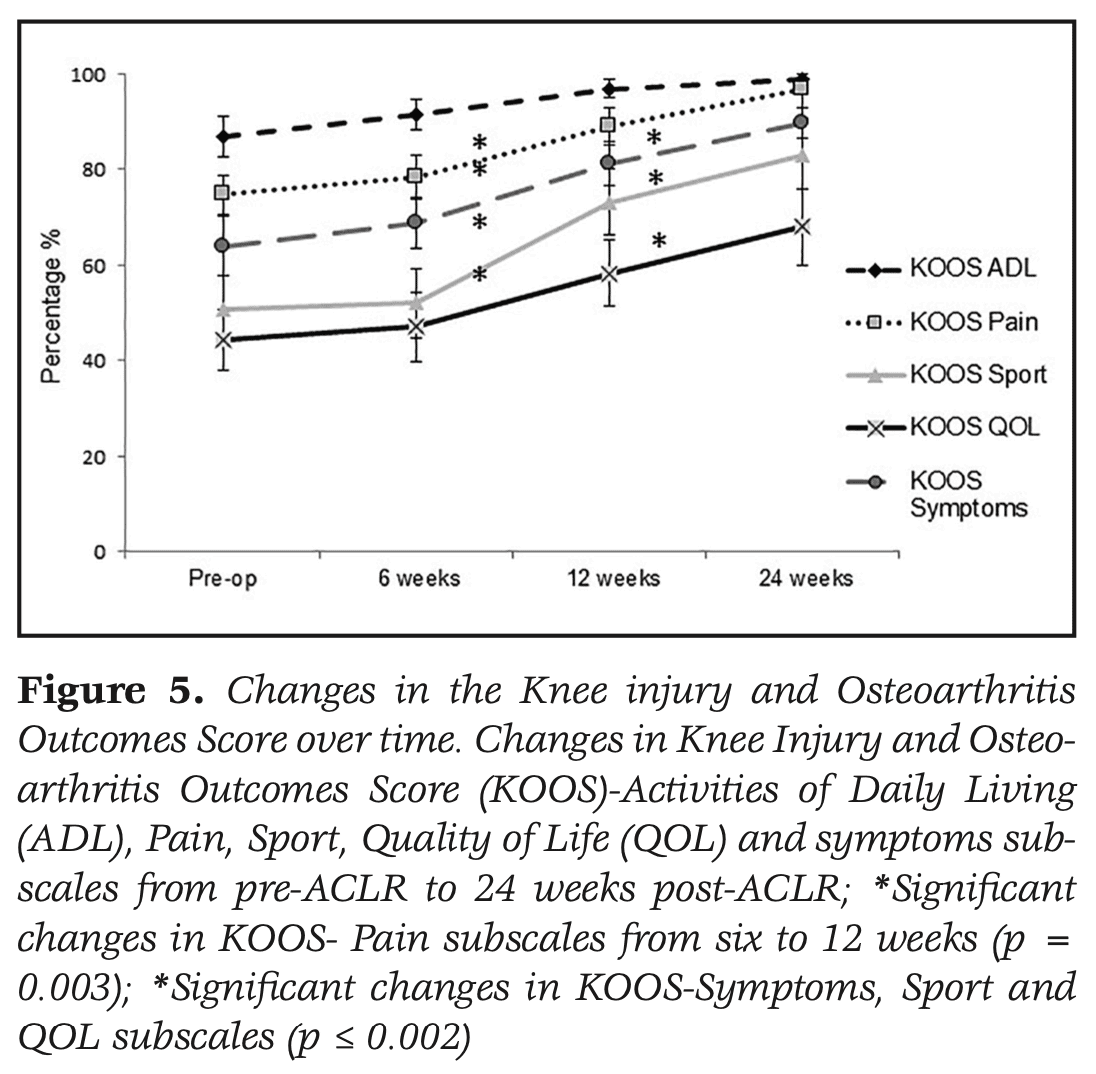
Knee-related function improved significantly over the course of the study. Significant improvements were seen between 6 and 12 weeks on the IKDC and KOOS pain, symptoms, QOL, and sport domains. From 12 to 24 weeks, significant improvements in the IKDC and KOOS symptoms, QOL, and Sport domains were observed.
Talk Nerdy to me
This study shows several good aspects. The sample size was a priori defined and the authors used a Bonferroni correction for multiple comparisons. All available data were included in the analyses, including data of those lost to follow-up. Over the course of the study, no significant changes in quadriceps peak torque of the uninvolved limb were seen, thus the reported improvements can be attributed to real gains in strength of the operated leg.
An important limitation of this study was the change of strength measurement procedure at 6 weeks as this may have caused why no significant improvements in strength have been noted at that time. Yet, in the light of ensuring maximal protection of the healing ACL graft, this deviation in the measurement method taken by the authors is understandable. Another limitation is the fact that at 24 weeks, only 44% of participants reached a QI of 90%, which may indicate that the rehabilitation efforts were insufficient.
Take home messages
Significant improvements in patient-reported knee function occurred 6 weeks post-ACLR, while improvements in quadriceps symmetry did not occur until the 12th week after ACLR. At 12 weeks, the mean QI was only 74% and therefore initiation of high-impact activities during this time frame was not appropriate for some athletes in this cohort. This once again shows the importance of obtaining adequate strength measures at different time points during rehabilitation.
Reference
LEARN TO OPTIMIZE REHAB & RTS DECISION MAKING AFTER ACL RECONSTRUCTION
Sign up for this FREE webinar and top leading expert in ACL rehab Bart Dingenen will show you exactly how you can do better in ACL rehab and return to sport decision making



