Effect of Mechanical Load on Tendon Adaptation - Biological Backgrounds for Precise Exercise Therapy Interventions
Introduksjon
Despite advances in our understanding of tendinopathy pathogenesis, outcomes following traditional conservative and medical interventions remain inconsistent, with many patients experiencing persistent pain and functional limitations. The widely cited continuum model proposed by Jill Cook suggests that the degenerative portion of a tendon is structurally irreversible. However, emerging structural and mechanobiological evidence challenges this assumption, indicating that tendon tissue may retain a greater capacity for adaptation and remodeling than previously thought.
This review explores the influence of mechanical load on tendon adaptation and the biological mechanisms underlying degeneration, with particular focus on cellular signaling, matrix remodeling, and mechanotransduction pathways. By integrating recent experimental findings, it aims to provide a biologically grounded framework to inform exercise prescription in tendinopathy management. This theoretical synthesis serves as the foundation for a forthcoming review examining high-load exercise strategies and introducing a novel tendinopathy loading protocol for clinical application.
Metoder
This narrative review synthesizes findings from multiple experimental studies, predominantly conducted in animal models.
Resultater
Biological Basis of Tendon Structure
Type I collagen (COL1/Col1a1) is the principal structural protein of tendon and ligament tissue, conferring tensile strength. Following injury, collagen synthesis increases; however, matrix deposition during the proliferative phase is often disorganized. Whereas healthy tendons exhibit collagen fibrils aligned parallel to mechanical forces, pathological tendons contain smaller, less cross-linked, and disorganized fibrils. Although type I collagen provides superior mechanical resistance compared with type III collagen, healing tendons frequently contain a higher proportion of type III collagen. Matrix metalloproteinases (MMPs), enzymes responsible for collagen degradation, are concurrently active during tissue remodeling.
Injured adult tendons typically become highly cellular and develop a disorganized collagen matrix characterized by small-diameter fibrils, resulting in a degenerative tissue phenotype.
Tissue Regeneration and Developmental Recapitulation
In response to injury, genes normally active during embryonic development are re-expressed. Stem and proliferative cell populations expand and differentiate into specialized tissue; however, adult tendons generally fail to fully regenerate, often leaving a residual degenerative matrix. In contrast, neonatal models demonstrate a greater capacity for functional repair, with tissue organization resembling developmental processes. These findings suggest that regenerative capacity may depend on the ability to recapitulate developmental programs, a feature that appears more robust in younger organisms and certain experimental models than in adult human tendon healing.
Material properties reflect mechanical demands
Tendons can be categorized according to their mechanical function as either energy-storing or positional structures. Energy-storing tendons, such as the Achilles tendon, absorb and return mechanical energy to enhance locomotor efficiency. Positional tendons, exemplified by the tibialis anterior tendon, primarily position joints and facilitate movements such as foot clearance during gait. These functional distinctions are reflected in structural properties: energy-storing tendons typically possess a larger cross-sectional area, compensating for lower material stiffness to enable elastic energy storage. Tendons within the same kinetic chain may also exhibit differing mechanical characteristics. For example, the quadriceps tendon and patellar tendon operate in series but display distinct stiffness properties, with the quadriceps tendon being approximately two times less stiff. This difference likely reflects their mechanical environments—bone-to-bone insertion for the patellar tendon versus muscle-to-bone attachment for the quadriceps tendon—which impose distinct loading patterns and therefore influence mechanical load on tendon adaptation and material requirements.
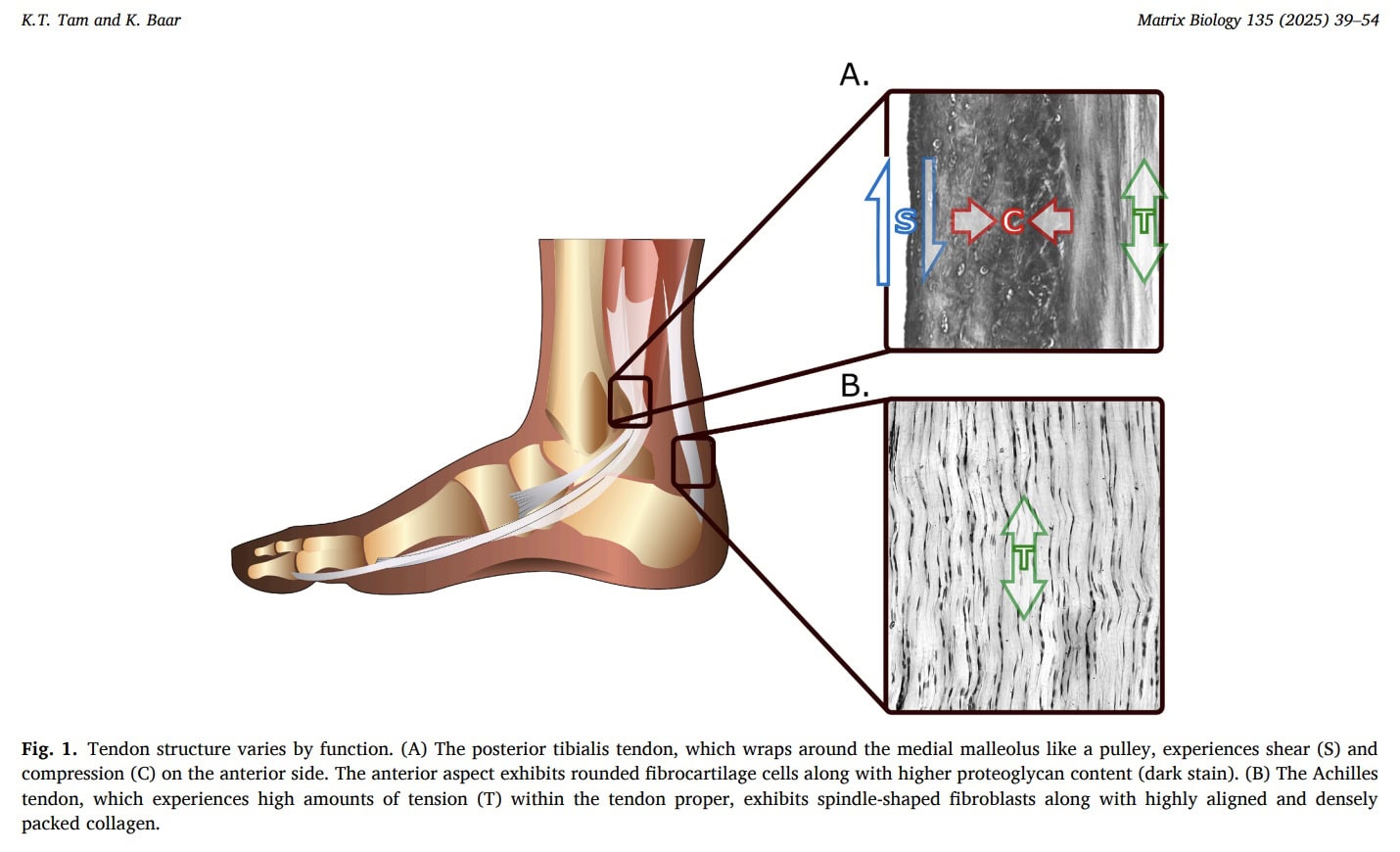
Compression
Compressive forces frequently act on tendons, particularly where they wrap around bony or retinacular structures. Experimental studies in animal models demonstrate that removal of compressive loading reduces tendon stiffness and alters structural adaptation, indicating that tendons exposed to compression remodel to accommodate this mechanical environment. In humans, compressed tendon regions often exhibit fibrocartilaginous features rich in type II collagen, specialized for resisting compressive forces.
Shear
Shear forces arise from relative sliding between tendons, ligaments, muscles, and adjacent tissues. In the Achilles tendon, differential sliding between fascicles facilitates force transmission but may diminish with age, potentially contributing to reduced range of motion and impaired force distribution. This decline in sliding capacity may partly explain the higher incidence of Achilles tendon rupture in older individuals. As with compressive adaptation, reduced mechanical loading may contribute to degenerative changes, increased injury risk, and desadaptation.
Specific forces drive specific molecular programs
Cell fate and tissue composition are strongly influenced by the mechanical environment through mechanotransduction pathways, emphasizing the importance of mechanical load on tendon adaptation. Mechanical forces initiate biological and metabolic adaptations that regulate tissue structure and function, although the specific pathways governing tendon healing remain incompletely understood. Subsequent sections examine the biomechanical effects of distinct loading modalities on tendon adaptation and repair.
Tension
Manipulation of the mechanical environment, including mechanical load, is central to tendon remodeling and functional adaptation. Tensile loading drives tissue remodeling, but the balance between beneficial and potentially harmful loads remains critical. A stronger biomechanical understanding of loading strategies is therefore essential to optimize tendon rehabilitation and adaptation.
Scleraxis (Scx) is a key transcription factor involved in tendon development and collagen regulation. During embryogenesis, Scx promotes type I collagen (COL1) synthesis by binding to regulatory regions of the Col1a1 gene. Its expression is influenced by muscle activity and mechanical loading. However, in adult tendons, Scx appears less essential for adaptive growth. Several tenogenic genes—including Col1a1, tenomodulin (Tnmd), fibromodulin (Fmod), and Mohawk (Mkx)—can be upregulated in response to load without corresponding changes in Scx expression. This suggests that tendon adaptation can occur independently of Scx and that its primary role may relate to early fibril formation rather than subsequent fibril growth. In contrast, Mkx appears to contribute to fibril enlargement and maturation in response to mechanical stimulation, supporting its role in tendon structural adaptation.
Healing adult tendons often exhibit features resembling developmental tissue, including small-diameter collagen fibrils and elevated Scx expression. However, unlike embryonic development, the healing matrix frequently fails to mature into organized, load-bearing tissue. One explanation is that mechanical signals may not be adequately transmitted through the scarred matrix, a phenomenon consistent with stress shielding (further discussed below). Impaired mechanical signalling could reduce activation of mechanosensitive pathways such as Mkx, limiting collagen fibril maturation and contributing to the formation of mechanically inferior scar tissue.
Compression
Compressive forces regulate tendon cell differentiation and matrix composition. Regions subjected to compression—such as the enthesis and tendon pulleys—commonly develop fibrocartilaginous features characterized by expression of cartilage markers including Col2a1 and aggrecan. Developmentally, tendon progenitors initially co-express Scx and Sox9 (a chondrogenic transcription factor) before segregating into tension-adapted tendon cells and compression-adapted fibrocartilage cells. Experimental evidence shows that sustained compression can induce cartilage-like tissue formation within the tendon, whereas tensile loading promotes tendon-specific gene expression and suppresses chondrogenic pathways. In the absence of Mkx, tensile strain may paradoxically favor chondrogenic gene expression, leading to ectopic fibrocartilage formation. These findings indicate that tendon cells possess multipotent potential and that mechanical loading governs differentiation through tension- and compression-sensitive transcriptional programs.
Shearing
Lubricin and hyaluronic acid are important mediators of tendon fascicle gliding and shear resistance. However, their biological regulation and response to mechanical loading remain insufficiently characterized, limiting understanding of their role in tendon pathology and adaptation.
Spatial arrangement
Collagen organization is essential for tendon function and is strongly regulated by mechanical tension. During development, tensile forces align cells and collagen fibrils through specialized structures (fibripositors), producing the parallel architecture characteristic of healthy tendons. Even in acellular collagen matrices, tensile strain can increase fibril alignment and density, and these changes may persist after unloading. However, the permanence of remodeling depends on matrix crosslinking, which may reduce adaptability in aging or metabolically altered tissues, such as diabetes. Tensile loading also increases resistance to collagen degradation and activates biochemical pathways (including Mkx) that support fibril maturation. Tendon adaptation, therefore, reflects the interaction of passive mechanical alignment and active cellular signalling in response to mechanical load, a process that governs structural remodelling and functional optimization.
Absent and aberrant forces play a role in tendon and ligament degeneration
Healing tendons often resemble immature or embryonic tissue, displaying elevated Scx expression, increased fibrillogenic collagens (III, V, XI), small-diameter collagen fibrils, high cellularity, vascularization, and the presence of Scx+/Sox9+ progenitors. These features suggest that the injured tendon reactivates a developmental program but fails to progress toward full mechanical maturation, likely due to altered mechanical signaling. Mechanical load is essential for proper tendon gene regulation: paralysis or unloading reduces key mechanosensitive transcription factors such as Egr1 and disrupts TGF-β–mediated Scx signaling, impairing regenerative capacity. Tensile loading promotes tendon-specific gene expression while suppressing cartilage genes, whereas compression or unloading shifts the balance toward chondrogenic or degenerative phenotypes. Although collagen III is commonly associated with scar tissue, evidence from regenerative models shows that its early upregulation is part of normal repair. Persistent elevation, particularly under unloading conditions, reflects failed maturation rather than the causation of degeneration. Even minimal mechanical strain is sufficient to regulate matrix gene expression and improve mechanical recovery, highlighting the extreme sensitivity of tendon cells to their loading environment. Together, these findings suggest that both absent and aberrant mechanical forces disrupt the normal progression from an early repair matrix toward a mature, mechanically competent tendon.
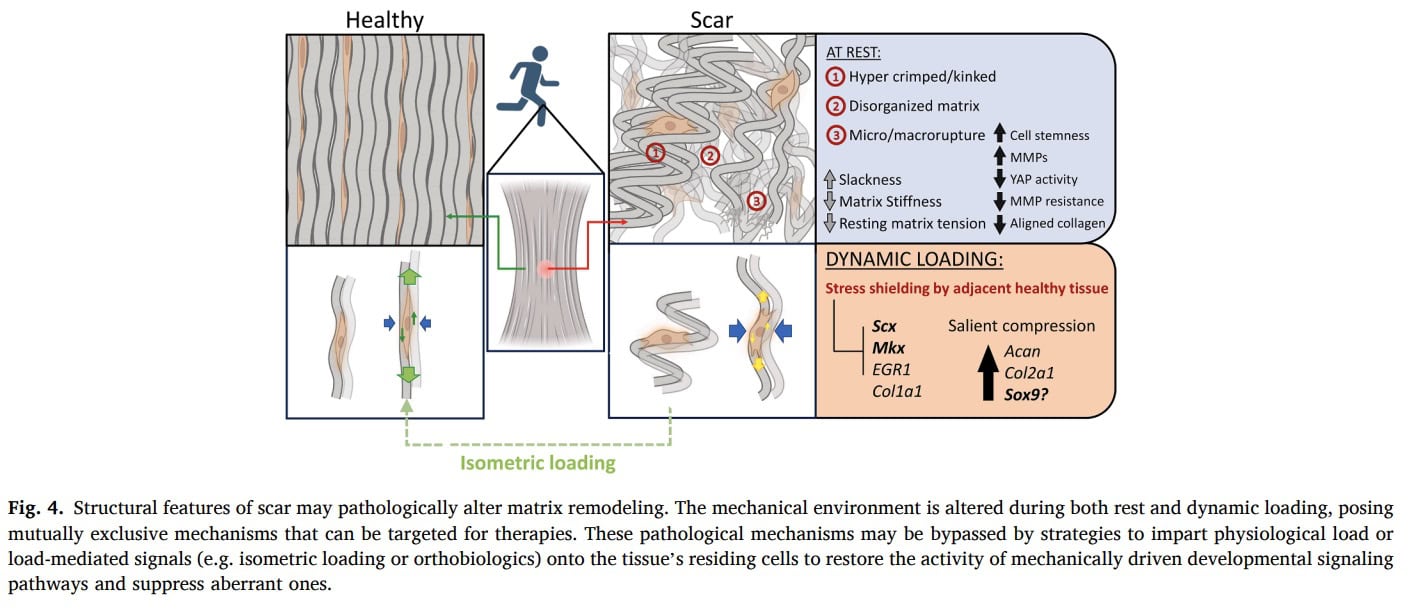
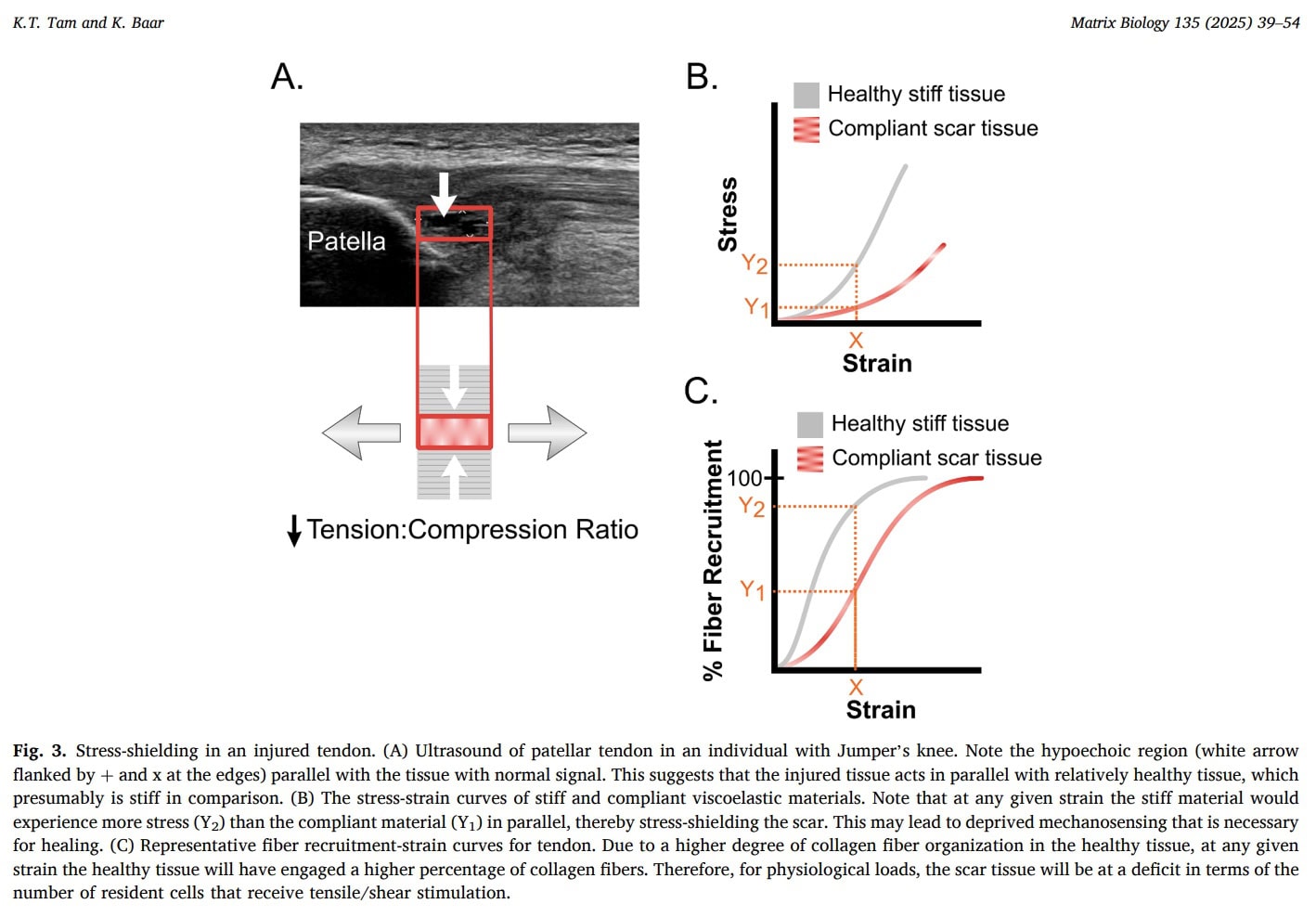
Stress shielding
As tendinopathy progresses, degenerative tendon regions may become symptomatic and subject to stress shielding. When mechanical load is applied, stiffer and healthier portions of the tendon preferentially bear the load, while more compliant degenerative regions are unloaded. This biomechanical phenomenon further reduces mechanical stimulation of the diseased tissue and may contribute to disuse and impaired remodeling. Because the degenerative portion receives little effective load, its capacity for tendon adaptation through mechanical load is diminished.
Tendons exhibit viscoelastic behavior, which can be leveraged therapeutically. Two key properties are stress relaxation—the gradual decrease in internal tension during a sustained stretch—and creep, the time-dependent deformation of tissue under constant stress. These properties suggest that controlled, sustained loading strategies may promote mechanical stimulation of degenerative regions despite stress shielding.
Isometric contractions may represent a valuable loading modality. Experimental models have demonstrated that isometric loading can upregulate tenogenic gene expression. Prolonged isometric contractions may induce stress relaxation in healthier tendon regions while permitting creep and mechanical strain within degenerative areas. This could facilitate load transmission to the scarred tissue and potentially support biological and structural adaptation.
Spørsmål og tanker
Tam et al. (2025) propose that in tendinopathy, compliant scar tissue may sit in parallel with stiffer healthy tendon and become mechanically “stress-shielded,” meaning that at normal physiological strain levels, fewer collagen fibers and resident cells within the scar actually experience tensile stress. Because key tenogenic regulators are load-sensitive, insufficient stress transmission may prevent scar maturation and instead favor persistence of an immature or fibrocartilage-like phenotype. For clinical practice, this model supports the rationale for carefully dosed mechanical loading rather than prolonged unloading: complete stress deprivation has been shown to impair tenogenic gene expression and mechanical recovery. The authors further suggest that sustained loading allowing viscoelastic creep (e.g., isometric contractions) may help transmit tension into the scar and activate tenogenic pathways, whereas inappropriate or absent loading may perpetuate degeneration. However, it is important to emphasize that while this mechanobiological framework provides a plausible explanation for why controlled loading could be beneficial and why fear of all loading may be misplaced, direct clinical evidence that specific loading strategies “overcome” stress-shielding in humans is not yet established within this paper.
Isometric loading is a promising modality for tendon rehabilitation, but optimal training parameters remain uncertain. In this case study, a combined isometric loading program and dietary supplementation protocol was applied to an athlete with chronic patellar tendinopathy. The dietary strategy consisted of 15 g of gelatin with 225 mg of vitamin C consumed approximately one hour before training sessions, aiming to support collagen synthesis.
The isometric exercise program targeted mid-range tendon loading using both open-chain (leg extension and leg press) and closed-chain (Spanish squat) exercises. Isometric holds were initially prescribed for 10 seconds and progressively increased by 5-second increments to a maximum of 30 seconds. Training volume ranged from one to three sets of two to four repetitions, performed at intensities exceeding 80% of one-repetition maximum (1 RM), which was recalculated monthly. Sessions lasted approximately 10 minutes, with hold durations chosen based on evidence showing that patellar tendon tension decreases by roughly 60% within 30 seconds of sustained contraction and only marginally thereafter.
Over the 18-month intervention, progressive increases in load and hold duration were associated with improvements in strength across all resisted tasks (leg extension, leg press, and Spanish squat holds). Magnetic resonance imaging (MRI) at baseline, 12 months, and 18 months demonstrated a reduction in tendon reactivity, increased tendon diameter at the midsubstance, and decreased thickness at the proximal insertion—findings consistent with structural remodeling. The athlete reported progressive pain reduction and was pain-free at the 18-month follow-up.
These observations suggest that isometric training, particularly when combined with nutritional strategies supporting collagen synthesis, may promote tendon adaptation and symptom improvement. However, evidence remains limited to experimental studies and isolated case reports. Larger controlled trials are required to determine the efficacy and optimal parameters of isometric protocols. Additionally, tendon properties vary according to anatomical location, cross-sectional area, and mechanical environment, which may influence viscoelastic behavior and appropriate loading strategies. As emphasized throughout this review, successful rehabilitation likely depends on achieving an appropriate balance between underloading and excessive loading, underscoring the need for reliable clinical tools to monitor mechanical load for tendon adaptation.
Snakk nerdete til meg
Although narrative reviews are subject to inherent biases such as selection bias, this review provides essential biological context that may help physiotherapists better understand tendon structure and mechanobiology in the management of tendinopathy. Knowledge of genetic regulation, transcriptional pathways, amino acid dynamics, and protein remodelling contributes to a deeper understanding of tendon healing and adaptation, which can inform rehabilitation strategies. However, most mechanistic evidence derives from animal models, and direct extrapolation to human tendon pathology remains limited.
While more advanced clinical trials are needed to strengthen the evidence base, the subsequent article in this series will examine data from a 2022 study investigating a high-load exercise protocol aimed at increasing tendon cross-sectional area and improving pain and function in patients with Achilles tendinopathy.
Ta med hjem meldinger
- Tendons are living, adaptive tissues. They respond to mechanical loading through cellular signaling and matrix remodeling. Mechanical stimuli drive structural adaptation—tendons are not inert structures.
- Biomechanical loading is essential for adaptation. Appropriate mechanical stress promotes tendon health and remodeling, supporting functional recovery and structural improvement through mechanical load on tendon adaptation.
- Stress shielding limits adaptation. Degenerative tendon regions may be unloaded when healthier tissue bears most of the mechanical load. This reduces effective mechanical stimulation and may hinder recovery.
- Rehabilitation must overcome stress shielding. Loading strategies should aim to transmit mechanical forces to degenerative tissue while avoiding excessive strain. Viscoelastic properties (stress relaxation and creep) provide a biomechanical basis for controlled, therapeutic loading.
- Balanced loading is key. Tendons require sufficient mechanical stimulus for adaptation but are vulnerable to underloading (which perpetuates disuse) and overloading (which may aggravate symptoms). Individualized, progressive loading is essential.
- Clinical implications for physiotherapy. Rehabilitation should focus on measurable and progressive loading strategies that restore mechanical stimulation to diseased tissue and harness tendon adaptability.
- Next steps. The upcoming article will translate these biomechanical principles into clinical strategies, reviewing high-load exercise protocols and practical approaches to optimize tendon adaptation, pain reduction, and functional recovery in tendinopathy.
This resource from Physiotutors provides additional perspectives on tendon biology and mechanobiology, offering clinically relevant insights into tendon function and adaptation.
Referanse
ROLLEN TIL VMO OG QUADS I PFP
Se denne GRATIS 2-DELS VIDEOFORSELSINGEN av knesmerterekspert Claire Robertson som dissekerer litteraturen om emnet og hvordan det påvirker klinisk praksis .